29 results
Sluzhenikinite, Pd15(Sb7-xSnx) 3 ≤ x ≤ 4, a new platinum group mineral (PGM) from the Oktyabrsk deposit, the Noril`sk deposits, Russia
- Part of
- Anna Vymazalová, Mark D. Welch, František Laufek, Vladimir V. Kozlov, Chris J. Stanley, Jakub Plášil
-
- Journal:
- Mineralogical Magazine / Volume 86 / Issue 4 / August 2022
- Published online by Cambridge University Press:
- 22 December 2021, pp. 577-585
-
- Article
- Export citation
-
Sluzhenikinite, Pd15(Sb7–xSnx) with 3 ≤ x ≤ 4, is a new mineral discovered in the pegmatoidal galena–chalcopyrite massive ore from the Oktyabrsk mine, Oktyabrsk deposit of the Noril`sk deposits, Russia. Sluzhenikinite forms euhedral elongate lamellar crystals (100–150 μm long and 10–50 μm wide) associated with Au–Ag alloy, insizwaite and myrmekitic intergrowths of Pt–Pd minerals (stibiopalladinite, maslovite and sobolevskite), in close association of sperrylite and base-metal sulfides (galena, chalcopyrite, cubanite and pentlandite). In plane-polarised light, sluzhenikinite is pale brown with weak bireflectance, imperceptible pleochroism, and weak anisotropy with straw yellow to deep blue rotation tints; it exhibits no internal reflections. Reflectance values for sluzhenikinite in air (R1,R2 in %) are: 46.2, 46.5 at 470nm; 52.1, 52.2 at 546nm; 54.7, 55.1 at 589nm; and 57.8, 59.0 at 650nm. Thirteen electron-microprobe analyses of sluzhenikinite gave an average composition: Pd 65.06, Sn 15.60 and Sb 19.58, total 100.24 wt.%, corresponding to the formula Pd14.88(Sb3.92Sn3.20)Σ7.12 based on 22 atoms; the average of twenty-one energy dispersive spectroscopy analyses on co-type material gave: Pd 63.36, Pt 1.15, Sn 16.28 and Sb 19.21, total 100.00 wt.%, corresponding to the formula (Pd14.62Pt0.14)Σ14.76(Sb3.87Sn3.37)Σ7.24. The density, calculated on the basis of the empirical formula, is 11.22 g/cm3. The mineral is monoclinic, space group P21/m, with a = 7.5558(1), b = 29.2967(3), c = 7.5713(1) Å, β = 119.931(2)°, V = 1452.44(4) Å3 and Z = 4. The crystal structure was determined using data from single-crystal X-ray diffraction and demonstrates conclusively that the correct stoichiometry is Pd15(Sb,Sn)7, rather than Pd2(Sb,Sn); R1 = 0.035, wR2 = 0.073, GoF = 1.118 for 209 refined parameters and 4738 unique reflections. The mineral is named after Sergey Fedorovich Sluzhenikin, an expert on platinum-group minerals, particularly from the area of the type locality.
Panskyite, Pd9Ag2Pb2S4, a new platinum group mineral from the Southern Kievey ore occurrence of the Fedorova–Pana layered intrusion, Kola Peninsula, Russia
- Anna Vymazalová, Viktor V. Subbotin, František Laufek, Yevgeny E. Savchenko, Chris J. Stanley, Dmitriy A. Gabov, Jakub Plášil
-
- Journal:
- Mineralogical Magazine / Volume 85 / Issue 2 / April 2021
- Published online by Cambridge University Press:
- 21 December 2020, pp. 161-171
-
- Article
- Export citation
-
Panskyite, Pd9Ag2Pb2S4, is a new mineral (IMA2020–039) discovered in the platinum-group element mineralisation of the Southern Kievey ore occurrence of the Fedorova–Pana layered intrusion, Kola Peninsula, Russia. It forms tiny anhedral grains (of 0.5 to 10 μm in size) in the interstices of rock-forming silicates, often forming tiny inclusions in base-metal sulfides (millerite, chalcopyrite, bornite and chalcocite) and complex intergrowths with other platinum group minerals (zvyagintsevite, laflammeite, vysotskite, thalhammerite, unnamed phase Pd9Ag2(Tl,Pb)2S4 and others). In plane-polarised light, panskyite is creamy white with weak bireflectance, weak pleochroism and distinct anisotropy with brown to grey rotation tints; it exhibits no internal reflections. Reflectance values for panskyite in air (R1, R2 in %) are: 43.8, 44.1 at 470 nm; 44.4, 44.7 at 546 nm; 45.6, 45.8 at 589 nm; and 47.2, 47.2 at 650 nm. Twelve electron-microprobe analyses of panskyite gave an average composition: Pd 55.61, Ag 12.36, Pb 23.50, Fe 0.21, Ni 0.24 and S 7.17 total 99.09 wt.%, corresponding to the formula (Pd9.05Fe0.07Ni0.07)Σ9.19Ag1.98Pb1.96S3.87 based on 17 atoms; the average of nine analyses on the synthetic analogue is: Pd 57.02, Ag 14.17, Pb 21.81 and S 7.44, total 100.44 wt.%, corresponding to Pd9.07Ag2.22Pb1.78S3.93. The density, calculated on the basis of the empirical formula, is 9.81 g/cm3. The mineral is tetragonal, space group I4/mmm, with a = 7.973(3), c = 9.139(3) Å, V = 581.0(4) Å3 and Z = 2. The crystal structure was solved from the single-crystal and powder X-ray diffraction data of synthetic Pd9Ag2Pb2S4. Panskyite is isostructural with thalhammerite (Pd9Ag2Bi2S4). The mineral name is for the locality, the Pansky massif of the Fedorova–Pana layered intrusion in the Kola Peninsula, Russia.
Mitrofanovite, Pt3Te4, a new mineral from the East Chuarvy deposit, Fedorovo–Pana intrusion, Kola Peninsula, Russia
- Victor V. Subbotin, Anna Vymazalová, František Laufek, Yevgeny E. Savchenko, Chris J. Stanley, Dmitry A. Gabov, Jakub Plášil
-
- Journal:
- Mineralogical Magazine / Volume 83 / Issue 4 / August 2019
- Published online by Cambridge University Press:
- 03 October 2018, pp. 523-530
-
- Article
- Export citation
-
Mitrofanovite, Pt3Te4, is a new telluride discovered in low-sulfide disseminated ore in the East Chuarvy deposit, Fedorovo–Pana intrusion, Kola Peninsula, Russia. It forms anhedral grains (up to ~20 μm × 50 μm) commonly in intergrowths with moncheite in aggregates with lukkulaisvaaraite, kotulskite, vysotskite, braggite, keithconnite, rustenburgite and Pt–Fe alloys hosted by a chalcopyrite–pentlandite–pyrrhotite matrix. Associated silicates are: orthopyroxene, augite, olivine, amphiboles and plagioclase. Mitrofanovite is brittle; it has a metallic lustre and a grey streak. Mitrofanovite has a good cleavage, along {001}. In plane-polarised light, mitrofanovite is bright white with medium to strong bireflectance, slight pleochroism, and strong anisotropy on non-basal sections with greyish brown rotation tints; it exhibits no internal reflections. Reflectance values for the synthetic analogue of mitrofanovite in air (Ro, Re’ in %) are: 58.4, 54.6 at 470 nm; 62.7, 58.0 at 546 nm; 63.4, 59.1 at 589 nm; and 63.6, 59.5 at 650 nm. Fifteen electron-microprobe analyses of mitrofanovite gave an average composition: Pt 52.08, Pd 0.19, Te 47.08 and Bi 0.91, total 100.27 wt.%, corresponding to the formula (Pt2.91Pd0.02)Σ2.93(Te4.02Bi0.05)Σ4.07 based on 7 atoms; the average of eleven analyses on synthetic analogue is: Pt 52.57 and Te 47.45, total 100.02 wt.%, corresponding to Pt2.94Te4.06. The density, calculated on the basis of the formula, is 11.18 g/cm3. The mineral is trigonal, space group R
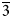 $\overline 3 $m, with a = 3.9874(1), c = 35.361(1) Å, V = 486.91(2) Å3 and Z = 3. The crystal structure was solved and refined from the powder X-ray-diffraction data of synthetic Pt3Te4. Mitrofanovite is structurally and chemically related to moncheite (PtTe2). The strongest lines in the powder X-ray diffraction pattern of synthetic mitrofanovite [d in Å (I) (hkl)] are: 11.790(23)(003), 5.891(100)(006), 2.851(26)(107), 2.137(16)(1013), 2.039(18)(0114), 1.574(24)(0120), 1.3098(21)(0027). The structural identity of natural mitrofanovite with synthetic Pt3Te4 was confirmed by electron backscatter diffraction measurements on the natural sample. The mineral name is chosen to honour Felix P. Mitrofanov, a Russian geologist who was among the first to discover platinum-group element mineralisation in the Fedorova–Pana complex.
$\overline 3 $m, with a = 3.9874(1), c = 35.361(1) Å, V = 486.91(2) Å3 and Z = 3. The crystal structure was solved and refined from the powder X-ray-diffraction data of synthetic Pt3Te4. Mitrofanovite is structurally and chemically related to moncheite (PtTe2). The strongest lines in the powder X-ray diffraction pattern of synthetic mitrofanovite [d in Å (I) (hkl)] are: 11.790(23)(003), 5.891(100)(006), 2.851(26)(107), 2.137(16)(1013), 2.039(18)(0114), 1.574(24)(0120), 1.3098(21)(0027). The structural identity of natural mitrofanovite with synthetic Pt3Te4 was confirmed by electron backscatter diffraction measurements on the natural sample. The mineral name is chosen to honour Felix P. Mitrofanov, a Russian geologist who was among the first to discover platinum-group element mineralisation in the Fedorova–Pana complex.
Klajite, MnCu4(AsO4)2(AsO3OH)2(H2O)10, from Jáchymov (Czech Republic): the second world occurrence
- J. Plášil, A. V. Kasatkin, R. Škoda, P. Škácha
-
- Journal:
- Mineralogical Magazine / Volume 78 / Issue 1 / February 2014
- Published online by Cambridge University Press:
- 05 July 2018, pp. 119-129
-
- Article
- Export citation
-
Klajite, MnCu4(AsO4)2(AsO3OH)2(H2O)10, the Mn-Cu-bearing member of the lindackerite group, was found in Jáchymov, Czech Republic, as the second world occurrence. It is associated with ondrušite and other arsenate minerals growing on the quartz gangue with disseminated primary sulfides, namely tennantite and chalcopyrite. Electron-microprobe data showed klajite aggregates to be chemically inhomogeneous at larger scales, varying from Mn-Ca-rich to Cu-rich domains. The chemical composition of the the Mn-rich parts of aggregates can be expressed by the empirical formula (Mn0.46Ca0.22Cu0.07Mg0.02)∑0.77(Cu3.82Mg0.14Ca0.03Zn0.01)∑4.00(As1.94Si0.06)∑2.00O8[AsO2.73(OH)1.27]2(H2O)10 (mean of seven representative spots; calculated on the basis of As + Si + P = 4 a.p.f.u. (atoms per formula unit) and 10 H2O from ideal stoichiometry), showing a slight cationic deficiency at the key Me-site. According to single-crystal X-ray diffraction, klajite from Jáchymov is triclinic, P
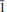 , with a = 6.4298(8), b = 7.9716(8), c = 10.707(2) Å, α = 85.737(12)°, β = 80.994(13)°, γ = 84.982(10)°, and V = 538.85(14) Å3, Z = 1. The crystal structure was refined to R1 = 0.0628 for 1034 unique observed reflections (with Iobs > 3σ(I)), confirming that klajite (Mn-Cu member) and ondrušite (Ca-Cu member) are isostructural. The current data-set allowed determination of the positions of several hydrogen atoms. Discussion on hydrogen bonding networks in the structure of klajite as well as detailed bond-valence analysis are provided.
, with a = 6.4298(8), b = 7.9716(8), c = 10.707(2) Å, α = 85.737(12)°, β = 80.994(13)°, γ = 84.982(10)°, and V = 538.85(14) Å3, Z = 1. The crystal structure was refined to R1 = 0.0628 for 1034 unique observed reflections (with Iobs > 3σ(I)), confirming that klajite (Mn-Cu member) and ondrušite (Ca-Cu member) are isostructural. The current data-set allowed determination of the positions of several hydrogen atoms. Discussion on hydrogen bonding networks in the structure of klajite as well as detailed bond-valence analysis are provided.
Crystal structure and formula revision of deliensite, Fe[(UO2)2(SO4)2(OH)2](H2O)7
- J. Plášil, J. Hauser, V. Petříček, N. Meisser, S. J. Mills, R. Škoda, K. Fejfarová, J. Čejka, J. Sejkora, J. Hloušek, J.-M. Johannet, V. Machovič, L. Lapčák
-
- Journal:
- Mineralogical Magazine / Volume 76 / Issue 7 / December 2012
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2837-2860
-
- Article
- Export citation
-
The crystal structure of deliensite, Fe[(UO2)2(SO4)2(OH)2](H2O)7, was solved by direct methods and refined to R1 = 6.24% for 5211 unique observed reflections [Iobs > 3σ(I)], on a crystal that was found to consist of rotational and inversion (merohedral) twins, from Jeroným mine, Abertamy in the Czech Republic. The presence of four twin domains was taken into account in the refinement. The structure is orthorhombic, space group Pnn2, with unit-cell parameters a = 15.8514(9), b = 16.2478(7), c = 6.8943(3) Å, V = 1775.6(1) Å3 and Z = 4. The crystal structure of deliensite contains uranyl-sulfate sheets with a phosphuranylite topology, consisting of dimers of edge-sharing uranyl pentagonal bipyramids linked by corner-sharing with sulfate tetrahedra. The sheets lie in the (100) plane and are decorated by [Fe2+O(H2O)5] octahedra; two weakly bonded H2O molecules are present in the interlayer. The [Fe2+O(H2O)5] octahedron is linked directly to the sheet via the uranyl oxygen atom. Adjacent sheets are linked by hydrogen bonds only. The sheet topology and geometrical isomerism is discussed and a comparison of the composition obtained from electron-probe microanalysis, powder-diffraction data, Raman and infrared spectra of deliensite samples from Mas d'Alary, Lodève, France; L'Ecarpière mine, Gétigné, France; and several localities at Jáchymov, Western Bohemia, Czech Republic is made.
Nestolaite, CaSeO3·H2O, a new mineral from the Little Eva mine, Grand County, Utah, USA
- A. V. Kasatkin, J. Plášil, J. Marty, A. A. Agakhanov, D. I. Belakovskiy, I. S. Lykova
-
- Journal:
- Mineralogical Magazine / Volume 78 / Issue 3 / June 2014
- Published online by Cambridge University Press:
- 05 July 2018, pp. 497-505
-
- Article
- Export citation
-
Nestolaite (IMA 2013-074), CaSeO3·H2O, is a new mineral species from the Little Eva mine, Grand County, Utah, USA. It is named in honour of the prominent Italian mineralogist and crystallographer Fabrizio Nestola. The new mineral was found on sandstone matrix as rounded aggregates up to 2 mm across and up to 0.05 μm thick consisting of tightly intergrown oblique-angled, flattened to acicular crystals up to 30 μm long and up to 7 μm (very rarely up to 15 μm) thick. Nestolaite associates with cobaltomenite, gypsum, metarossite, orschallite and rossite. The new mineral is light violet and transparent with a white streak and vitreous lustre. The Mohs hardness is 2½. Nestolaite is brittle, has uneven fracture and perfect cleavage on {100}. The measured and calculated densities are Dmeas. = 3.18(2) g/cm3 and Dcalc. = 3.163 g/cm3. Optically, nestolaite is biaxial positive. The refractive indices are α = 1.642(3), β = 1.656(3), γ = 1.722(6). The measured 2V is 55(5)° and the calculated 2V is 51°. In transmitted light nestolaite is colourless. It does not show pleochroism but has strong pseudoabsorption caused by high birefringence. The chemical composition of nestolaite (wt.%, electronmicroprobe data) is: CaO 28.97, SeO2 61.14, H2O (calc.) 9.75, total 99.86. The empirical formula calculated on the basis of 4 O a.p.f.u. (atoms per formula unit) is Ca0.96Se1.02O3·H2O. The Raman spectrum is dominated by the Se–O stretching and O–Se–O bending vibrations of the pyramidal SeO3 groups and O–H stretching modes of the H2O molecules. The mineral is monoclinic, space group P21/c, with a = 7.6502(9), b = 6.7473(10), c = 7.9358(13) Å, β = 108.542 (12)°, V = 388.37(10) Å3 and Z = 4. The eight strongest powder X-ray diffraction lines are [dobs in Å(hkl) (Irel)]: 7.277 (100)(100), 4.949 (110)(37), 3.767 (002)(29), 3.630 (200)(58), 3.371 (020)(24), 3.163 (
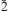 02)(74), 2.9783 (
02)(74), 2.9783 (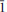 21)(74) and 2.7231 (112)(31). The crystal structure of nestolaite was determined by means of the Rietveld refinement from the powder data to Rwp = 0.019. Nestolaite possesses a layered structure consisting of CaΦ–SeO3 sheets, composed of edge-sharing polyhedra. Adjacent sheets are held by H bonds emanating from the single (H2O) group within the sheets. The nestolaite structure is topologically unique.
21)(74) and 2.7231 (112)(31). The crystal structure of nestolaite was determined by means of the Rietveld refinement from the powder data to Rwp = 0.019. Nestolaite possesses a layered structure consisting of CaΦ–SeO3 sheets, composed of edge-sharing polyhedra. Adjacent sheets are held by H bonds emanating from the single (H2O) group within the sheets. The nestolaite structure is topologically unique.
Štěpite, U(AsO3OH)2·4H2O, from Jáchymov, Czech Republic: the first natural arsenate of tetravalent uranium
- J. Plášil, K. Fejfarová, J. Hloušek, R. Škoda, M. Novák, J. Sejkora, J. Čejka, M. Dušek, F. Veselovský, P. Ondruš, J. Majzlan, Z. Mrázek
-
- Journal:
- Mineralogical Magazine / Volume 77 / Issue 1 / February 2013
- Published online by Cambridge University Press:
- 05 July 2018, pp. 137-152
-
- Article
- Export citation
-
Štěpite, tetragonal U(AsO3 OH)2(H2O)4 (IMA 2012-006), is the first natural arsenate of tetravalent uranium. It occurs in the Geschieber vein, Jáchymov ore district, Western Bohemia, Czech Republic, as emerald-green crystalline crusts on altered arsenic. Associated minerals include arsenolite, běhounekite, claudetite, gypsum, kaatialaite, the new mineral vysokýite (IMA 2012-067) and a partially characterized phase with the formula (H3O)+2(UO2)2(AsO4)2˙6H2O. Štěpite typically forms tabular crystals with prominent {001} and {010} faces, up to 0.6 mm in size. The crystals have a vitreous lustre and a grey to greenish grey streak. They are brittle with an uneven fracture and a very good cleavage on (001). Their Mohs hardness is about 2. Štěpite is not fluorescent in either short-wave or long-wave ultraviolet light. It is biaxial (–) with refractive indices (at 590 nm) of α = 1.636(2), β = 1.667(3), γ = 1.672(2) and 2Vobs < ~5°, anomalous greyish to pale yellow interference colours, and no pleochroism. The composition is as follows: 0.12Na2O, 50.19 UO2, 0.04SiO4, 0.09 P2O5, 0.93 As2O5, 1.95 SO3, 16.41 H2O; total 107.90 wt.%, yielding an empirical formula (based on 12 O a. p. f. u.) of (U1.01Na0.02)Σ1.03[(AsO3OH)1.82 (PO3OH)0.04(SO4)0.13(SiO4)0.01]Σ 2.00˙4H2O. Štěpite is tetragonal, crystallizing in space group I41/acd, with a = 10.9894(1), c = 32.9109(6) Å, V = 3974.5(1) Å3, Z = 16 and Dcalc = 3.90 g cm-3. The six strongest peaks in the X-ray powder-diffraction pattern [dobs in Å (I) (hkl)] are as follows: 8.190(100)(004), 7.008(43)(112), 5.475(18)(200), 4.111(16)(008), 3.395(20)(312,217), 2.1543(25)(419). The crystal structure of šteěpite has been solved from singlecrystal X-ray diffraction data by the charge-flipping method and refined to R1 = 0.0353 based on 1434 unique observed reflections, and to wR2 = 0.1488 for all 1523 unique reflections. The crystal structure of štšpite consists of sheets perpendicular to [001], made up of eight-coordinate uranium atoms and hydroxyarsenate 'tetrahedra'. The ligands surrounding the uranium atom consist of six oxygen atoms which belong to the hydroxyarsenate groups and two oxygen atoms which belong to interlayer H2 O molecules. Each UO8 polyhedron is connected to five other U polyhedra via six AsO3OH groups. Adjacent electroneutral sheets, of composition [U4+(AsO3OH)22-]0, are linked by hydrogen bonds involving H2 O molecules in the interlayers and OH groups in the sheets. The new mineral is named in honour of Josef Štěp (1863–1926), a Czech mining engineer and 'father' of the world's first radioactive spa at Jáchymov.
Widenmannite, a rare uranyl lead carbonate: occurrence, formation and characterization
- J. Plášil, J. Čejka, J. Sejkora, P. Škácha, V. Goliáš, P. Jarka, F. Laufek, J. Jehlička, I. Němec, L. Strnad
-
- Journal:
- Mineralogical Magazine / Volume 74 / Issue 1 / February 2010
- Published online by Cambridge University Press:
- 05 July 2018, pp. 97-110
-
- Article
- Export citation
-
The rare uranyl lead carbonate widenmannite, Pb2(UO2)(CO3)3, was found at the Jánská vein, Příbram, Czech Republic, where two generations occur in several morphological types and mineral associations in hydrothermal veins. Alpha spectroscopy shows that these two generations have different ages, >220,000 and 118±12 y. ICP-MS analysis indicates that both widenmannites have a dominance of non-radiogenic Pb which originates from weathered galena. The older widenmannite I forms fine-grained, grey to beige aggregates in the highly altered supergene part of the hydrothermal ore vein in association with pyromorphite, cerussite and goethite. The younger widenmannite II occurs as white, yellow or greenish-yellow thin tabular crystals upto 0.5 mm long in association with cerussite, anglesite, limonite, kasolite and an unnamed Pb-U-O phase. Thermal analysis suggests that widenmannite decomposes in several steps, with Pb uranate as the final product. Infrared and Raman spectroscopy confirm the presence of non-equivalent (CO3)2– groups, bidentately coordinated in uranyl hexagonal polyhedra, forming the well known uranyl tricarbonate complex. Infrared spectroscopy shows conclusively that widenmannite does not contain molecular H2O.
The crystal structure of rabejacite, the Ca2+-dominant member of the zippeite group
- J. Plášil, M. Dušek, J. Čejka, J. Sejkora
-
- Journal:
- Mineralogical Magazine / Volume 78 / Issue 5 / October 2014
- Published online by Cambridge University Press:
- 05 July 2018, pp. 1249-1264
-
- Article
- Export citation
-
The crystal structure of rabejacite from Jáchymov, ideally Ca2[(UO2)4O4(SO4)2](H2O)8, was solved by charge flipping from single-crystal data and refined to R1 = 11.94% for 1422 unique observed reflections [I > 3σ(I)]. According to single-crystal X-ray data, rabejacite is triclinic, space group P
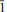 , with a = 8.7434(11), b = 8.309(3), c = 8.8693(10) Å , a = 77.86(2), b = 104.635(11), g = 82.935(18)°, V = 598.8(3) A˚ 3 and Z = 1, with Dcalc = 4.325 g cm–3. The structure refinement proved that rabejacite is related to the zippeite group of minerals, as it is based upon the structural sheets of the zippeite topology of composition [(UO2)4O4(SO4)2]4–. Located in the interlayer between the sheets, which are stacked perpendicular to [010], are Ca2+ cations and H2O groups. Ca2+ ions are [7]-coordinated, by three uranyl O atoms from adjacent sheets and four H2O groups. An additional H2O group, which is not bonded directly to any cation, is located in the interlayer. Along with rabejacite, its Cu-rich variety was found in the specimens examined and characterized structurally. Its crystal structure (R1 = 10.15% for 1049 reflections with I > 3s(I)) is practically the same as that of rabejacite, but there is an additional Cu2+ site located in between pairs of Ca polyhedra. The structural formula is (Ca1.56Cu0.40)Σ1.90[(UO2)4O4(SO4)2](H2O)8, Z = 1. Its existence suggests a greater diversity in zippeite crystal chemistry than was thought previously and also the possibility of a new Cu2+-dominant zippeite mineral besides pseudojohannite.
, with a = 8.7434(11), b = 8.309(3), c = 8.8693(10) Å , a = 77.86(2), b = 104.635(11), g = 82.935(18)°, V = 598.8(3) A˚ 3 and Z = 1, with Dcalc = 4.325 g cm–3. The structure refinement proved that rabejacite is related to the zippeite group of minerals, as it is based upon the structural sheets of the zippeite topology of composition [(UO2)4O4(SO4)2]4–. Located in the interlayer between the sheets, which are stacked perpendicular to [010], are Ca2+ cations and H2O groups. Ca2+ ions are [7]-coordinated, by three uranyl O atoms from adjacent sheets and four H2O groups. An additional H2O group, which is not bonded directly to any cation, is located in the interlayer. Along with rabejacite, its Cu-rich variety was found in the specimens examined and characterized structurally. Its crystal structure (R1 = 10.15% for 1049 reflections with I > 3s(I)) is practically the same as that of rabejacite, but there is an additional Cu2+ site located in between pairs of Ca polyhedra. The structural formula is (Ca1.56Cu0.40)Σ1.90[(UO2)4O4(SO4)2](H2O)8, Z = 1. Its existence suggests a greater diversity in zippeite crystal chemistry than was thought previously and also the possibility of a new Cu2+-dominant zippeite mineral besides pseudojohannite.
Švenekite, Ca[AsO2(OH)2]2, a new mineral from Jáchymov, Czech Republic
- P. Ondruš, R. Skála, J. Plášil, J. Sejkora, F. Veselovský, J. Čejka, A. Kallistová, J. Hloušek, K. Fejfarová, R. Škoda, M. Dušek, A. Gabašová, V. Machovič, L. Lapčák
-
- Journal:
- Mineralogical Magazine / Volume 77 / Issue 6 / August 2013
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2711-2724
-
- Article
- Export citation
-
Švenekite (IMA 99-007), Ca[AsO2(OH)2]2, is a rare supergene arsenate mineral occurring in the Geschieber vein, Jáchymov ore district, Western Bohemia, Czech Republic. It grows directly on the granite rocks and occurs isolated from other arsenate minerals otherwise common in Jáchymov. Švenekite usually forms clear transparent coatings composed of indistinct radiating to rosette-shaped aggregates up to 3 mm across. They are composed of thin lens- or bladed-shaped crystals, usually 100 – 150 μm long. Švenekite is transparent to translucent and has a white streak and a vitreous lustre; it does not fluoresce under ultraviolet light. Cleavage is very good on {010}. The Mohs hardness is ∼2. Švenekite is biaxial, non-pleochroic. The refractive indices are α' = 1.602(2), γ' = 1.658(2). The empirical formula of švenekite (based on As + P + S = 2 a.p.f.u., an average of 10 spot analyses) is (Ca1.00Mg0.01)Σ1.01[AsO2(OH)2]1.96[PO2(OH)2]0.03(SO4)0.01. The simplified formula is Ca[AsO2(OH)2]2 and requires CaO 17.42, As2O571.39, H2O 11.19, total 100.00 wt.%. Raman and infrared spectroscopy exhibit dominance of O – H vibrations and vibration modes of distorted tetrahedral AsO2(OH)2 units. Švenekite is triclinic, space group P
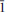 , with a = 8.5606(5), b = 7.6926(6), c = 5.7206(4) Å, α = 92.605(6), β = 109.9002(6), γ = 109.9017(6)º, and V = 327.48(4) Å3, Z = 2, Dcalc = 3.26 g·cm–3. The a:b:c ratio is 0.7436:1:1.1082 (for single-crystal data). The six strongest diffraction peaks in the X-ray powder diffraction pattern are [d (Å)/I(%)/(hkl)]: 3.968(33)(2
, with a = 8.5606(5), b = 7.6926(6), c = 5.7206(4) Å, α = 92.605(6), β = 109.9002(6), γ = 109.9017(6)º, and V = 327.48(4) Å3, Z = 2, Dcalc = 3.26 g·cm–3. The a:b:c ratio is 0.7436:1:1.1082 (for single-crystal data). The six strongest diffraction peaks in the X-ray powder diffraction pattern are [d (Å)/I(%)/(hkl)]: 3.968(33)(2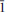 0); 3.766(35)(2
0); 3.766(35)(2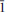
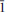 ); 3.697(49)(101); 3.554(100)(020); 3.259(33)(2
); 3.697(49)(101); 3.554(100)(020); 3.259(33)(2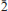 0); 3.097(49)(1
0); 3.097(49)(1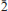 1). The crystal structure of švenekite was refined from single-crystal X-ray diffraction data to R1 = 0.0250 based on 1309 unique observed, and to wR2 = 0.0588, for all 1588 unique reflections (with GOFall = 1.20). The structure of švenekite consists of sheets of corner-sharing CaO8 polyhedra and AsO2 OH2 groups, stacked parallel to (001). Adjacent sheets are linked by hydrogen bonds. The švenekite structure possesses very short symmetrical hydrogen bonds (with the D–H lengths ∼1.22 Å). The mineral is named to honour Jaroslav Švenek, the former curator of the mineralogical collection of the National Museum in Prague, Czech Republic.
1). The crystal structure of švenekite was refined from single-crystal X-ray diffraction data to R1 = 0.0250 based on 1309 unique observed, and to wR2 = 0.0588, for all 1588 unique reflections (with GOFall = 1.20). The structure of švenekite consists of sheets of corner-sharing CaO8 polyhedra and AsO2 OH2 groups, stacked parallel to (001). Adjacent sheets are linked by hydrogen bonds. The švenekite structure possesses very short symmetrical hydrogen bonds (with the D–H lengths ∼1.22 Å). The mineral is named to honour Jaroslav Švenek, the former curator of the mineralogical collection of the National Museum in Prague, Czech Republic.
Manganoblödite, Na2Mn(SO4)2·4H2O, and cobaltoblödite, Na2Co(SO4)2·4H2O: two new members of the blödite group from the Blue Lizard mine, San Juan County, Utah, USA
- A. V. Kasatkin, F. Nestola, J. Plášil, J. Marty, D. I. Belakovskiy, A. A. Agakhanov, S. J. Mills, D. Pedron, A. Lanza, M. Favaro, S. Bianchin, I. S. Lykova, V. Goliáš, W. D. Birch
-
- Journal:
- Mineralogical Magazine / Volume 77 / Issue 3 / April 2013
- Published online by Cambridge University Press:
- 05 July 2018, pp. 367-383
-
- Article
- Export citation
-
Two new minerals – manganoblödite (IMA2012–029), ideally Na2Mn(SO4)2·4H2O, and cobaltoblödite (IMA2012–059), ideally Na2Co(SO4)2·4H2O, the Mn-dominant and Co-dominant analogues of blödite, respectively, were found at the Blue Lizard mine, San Juan County, Utah, USA. They are closely associated with blödite (Mn-Co-Ni-bearing), chalcanthite, gypsum, sideronatrite, johannite, quartz and feldspar. Both new minerals occur as aggregates of anhedral grains up to 60 μm (manganoblödite) and 200 μm (cobaltoblödite) forming thin crusts covering areas up to 2 × 2 cm on the surface of other sulfates. Both new species often occur as intimate intergrowths with each other and also with Mn-Co-Ni-bearing blödite. Manganoblödite and cobaltoblödite are transparent, colourless in single grains and reddish-pink in aggregates and crusts, with a white streak and vitreous lustre. Their Mohs' hardness is ∼2½. They are brittle, have uneven fracture and no obvious parting or cleavage. The measured and calculated densities are Dmeas = 2.25(2) g cm−3 and Dcalc = 2.338 g cm−3 for manganoblödite and Dmeas = 2.29(2) g cm−3 and Dcalc = 2.347 g cm−3 for cobaltoblödite. Optically both species are biaxial negative. The mean refractive indices are α = 1.493(2), β = 1.498(2) and γ = 1.501(2) for manganoblödite and α = 1.498(2), β = 1.503(2) and γ = 1.505(2) for cobaltoblödite. The chemical composition of manganoblödite (wt.%, electron-microprobe data) is: Na2O 16.94, MgO 3.29, MnO 8.80, CoO 2.96, NiO 1.34, SO3 45.39, H2O (calc.) 20.14, total 98.86. The empirical formula, calculated on the basis of 12 O a.p.f.u., is: Na1.96(Mn0.44Mg0.29Co0.14Ni0.06)Σ0.93S2.03O8·4H2O. The chemical composition of cobaltoblödite (wt.%, electron-microprobe data) is: Na2O 17.00, MgO 3.42, MnO 3.38, CoO 7.52, NiO 2.53, SO3 45.41, H2O (calc.) 20.20, total 99.46. The empirical formula, calculated on the basis of 12 O a.p.f.u., is: Na1.96(Co0.36Mg0.30Mn0.17Ni0.12)Σ 0.95S2.02O8·4H2O. Both minerals are monoclinic, space group P21/a, with a = 11.137(2), b = 8.279(1), c = 5.5381(9) Å, β = 100.42(1)°, V = 502.20(14) Å3 and Z = 2 (manganoblödite); and a = 11.147(1), b = 8.268(1), C = 5.5396(7) Å, β = 100.517(11)°, V = 501.97(10) Å3 and Z = 2 (cobaltoblödite). The strongest diffractions from X-ray powder pattern [listed as (d,Å(I)(hkl)] are for manganoblödite: 4.556(70)(210, 011); 4.266(45)(
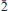 01); 3.791(26)(
01); 3.791(26)(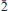 11); 3.338(21)(310); 3.291(100)(220, 021), 3.256(67)(211,
11); 3.338(21)(310); 3.291(100)(220, 021), 3.256(67)(211, 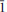 21), 2.968(22)(
21), 2.968(22)(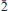 21), 2.647(24)(
21), 2.647(24)(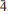 01); for cobaltoblödite: 4.551(80)(210, 011); 4.269(50)(
01); for cobaltoblödite: 4.551(80)(210, 011); 4.269(50)(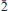 01); 3.795(18)(
01); 3.795(18)(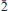 11); 3.339(43)(310); 3.29(100)(220, 021), 3.258(58)(
11); 3.339(43)(310); 3.29(100)(220, 021), 3.258(58)(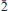 11,
11, 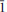 21), 2.644(21)(
21), 2.644(21)(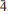 01), 2.296(22)(122). The crystal structures of both minerals were refined by single-crystal X-ray diffraction to R1 = 0.0459 (manganoblödite) and R1 = 0.0339 (cobaltoblödite).
01), 2.296(22)(122). The crystal structures of both minerals were refined by single-crystal X-ray diffraction to R1 = 0.0459 (manganoblödite) and R1 = 0.0339 (cobaltoblödite).
Iangreyite: a new secondary phosphate mineral closely related to perhamite
- S. J. Mills, A. R. Kampf, J. Sejkora, P. M. Adams, W. D. Birch, J. Plášil
-
- Journal:
- Mineralogical Magazine / Volume 75 / Issue 2 / April 2011
- Published online by Cambridge University Press:
- 05 July 2018, pp. 327-336
-
- Article
- Export citation
-
Iangreyite, ideally Ca2Al7(PO4)2(PO3OH)2(OH,F)15·8H2O, is a new mineral (IMA2009-087) from the Silver Coin mine, Nevada, USA and the Krásno ore district, Horní Slavkov, Czech Republic. At Silver Coin, iangreyite occurs as thin, colourless to white or cream, hexagonal tablets up to 0.4 mm in diameter and 0.02 mm thick associated with meurigite-Na, plumbogummite, kidwellite, lipscombite. strengite, chalcosiderite, wardite, leucophosphite, wavellite, goethite, barite, quartz and F-rich perhamite. At Krásno, white, yellowish or light pink iangreyite coatings consist of 0.3 mm wide clusters of minute and very thin intergrown tabular crystals with a maximum diameter of 0.2 mm. Individual iangreyite crystals are transparent with a vitreous lustre, while iangreyite clusters tend to be pearly and translucent. The estimated hardness is 3 on the Mohs scale, the fracture is irregular and the mineral is non-fluorescent under SW and LW ultraviolet light. Individual crystals are somewhat flexible and there is perfect cleavage on ﹛001﹜. The density (Silver Coin), measured by the sink-float method in an aqueous solution of sodium polytungstate, is 2.46(3) g/cm3, while the calculated density is 2.451 g/cm3. Crystals from Silver Coin are uniaxial (+), with the indices of refraction: ε = 1.544(2) and s = 1.554(2), measured in white light, and are non-pleochroic. The empirical formula for iangreyite from Silver Coin (calculated on the basis of 39 anions per formula unit) is: Ca1.42K0.22Na0.09Ba0.03 Sr0.01Al6.51Mg0.09Fe0.02Cu0.01Zn0.01P3.81F5.24H30.21O33.76, while the empirical formula from Krásno is: Ca2.15K0.10Na0.01Ba0.02Sr0.12Al6.28Mg0.01Fe0.12Cu0.08Zn0.01P3.64Si0.43F4.65H29.62O34.35. Iangreyite is trigonal, space group P321 and Z = 1, with the unit-cell parameters (Silver Coin): a = 6.988(1), c = 16.707(3) Å and V= 706.5(2) Å3 and (Krasno): a = 6.989(1), c = 16.782(4) Å and V = 709.8(2) Å3. The structure of iangreyite, modelled from powder data, consists of blocks of the crandallite-type structure that are interconnected along c via corner-sharing of crandallite-block PO4 tetrahedra with A1O2(OH)3 bipyramids. This linkage generates large channels along [110] bounded by 10-member rings of octahedra, tetrahedra and trigonal biyramids, that are occupied by Ca and water molecules.
Hydroniumjarosite, (H3O)+Fe3(SO4)2(OH)6, from Cerros Pintados, Chile: Single-crystal X-ray diffraction and vibrational spectroscopic study
- J. Plášil, R. Škoda, K. Fejfarová, J. Čejka, A. V. Kasatkin, M. Dušek, D. Talla, L. Lapčák, V. Machovič, M. Dini
-
- Journal:
- Mineralogical Magazine / Volume 78 / Issue 3 / June 2014
- Published online by Cambridge University Press:
- 05 July 2018, pp. 535-547
-
- Article
- Export citation
-
The natural hydroniumjarosite sample from Cerros Pintados (Chile) was investigated by electron microprobe, single-crystal X-ray diffraction and vibrational spectroscopy (Infrared and Raman). The chemical composition of studied specimens (wt.%, mean of seven analyses) obtained from electron microprobe (in wt.%): Na2O 1.30, K2O 0.23, CaO 0.04, Fe2O3 50.49, Al2O3 0.37, SiO2 0.33, SO3 33.88, H2O (calculated on the basis of Σ(OH–+H3O+) deduced from the charge balance) 13.32, total 99.98, corresponds to the empirical formula (H3O)0.77+(Na0.20K0.02)∑0.22(Fe2.95Al0.03)∑2.98 (OH)6.12[(SO4)1.97(SiO4)0.03]∑2.00 (calculated on the basis of S + Si = 2 a.p.f.u. (atoms per formula unit)). The studied hydroniumjarosite is trigonal, with space group R
 m, with a = 7.3408(2), c = 17.0451(6) Å and V = 795.46(4) Å3. The refined structure architecture is consistent with known jarosite-series minerals, including synthetic hydroniumjarosite. However, in the current study the presence of H3O+ is well documented in difference Fourier maps, where characteristic positive difference Fourier maxima, with apparent trigonal symmetry, were localized in the vicinity of the O4 atom in the channel-voids of the structure. The structure of natural hydroniumjarosite, including the H atoms, was refined to R1 = 0.0166 for 2113 unique observed reflections, with Iobs>3σ(I). The present structure model, which includes the position of the H atom within the hydronium ion, is discussed with regard to the vibration spectroscopy results and earlier published density-functional theory (DFT) calculations for the alunite-like structure containing H3O+.
m, with a = 7.3408(2), c = 17.0451(6) Å and V = 795.46(4) Å3. The refined structure architecture is consistent with known jarosite-series minerals, including synthetic hydroniumjarosite. However, in the current study the presence of H3O+ is well documented in difference Fourier maps, where characteristic positive difference Fourier maxima, with apparent trigonal symmetry, were localized in the vicinity of the O4 atom in the channel-voids of the structure. The structure of natural hydroniumjarosite, including the H atoms, was refined to R1 = 0.0166 for 2113 unique observed reflections, with Iobs>3σ(I). The present structure model, which includes the position of the H atom within the hydronium ion, is discussed with regard to the vibration spectroscopy results and earlier published density-functional theory (DFT) calculations for the alunite-like structure containing H3O+.
Belakovskiite, Na7(UO2)(SO4)4(SO3OH)(H2O)3, a new uranyl sulfate mineral from the Blue Lizard mine, San Juan County, Utah, USA
- A. R. Kampf, J. Plášil, A. V. Kasatkin, J. Marty
-
- Journal:
- Mineralogical Magazine / Volume 78 / Issue 3 / June 2014
- Published online by Cambridge University Press:
- 05 July 2018, pp. 639-649
-
- Article
- Export citation
-
The new mineral belakovskiite (IMA2013-075), Na7(UO2)(SO4)4(SO3OH)(H2O)3, was found in the Blue Lizard mine, Red Canyon, White Canyon district, San Juan County, Utah, USA, where it occurs as a secondary alteration phase in association with blödite, ferrinatrite, kröhnkite, meisserite and metavoltine. Crystals of belakovskiite are very pale yellowish-green hair-like fibres up to 2 mm long and usually no more than a few mm in diameter. The fibres are elongated on [100] and slightly flattened on {021}. Crystals are transparent with a vitreous lustre. The mineral has a white streak and a probable Mohs hardness of ∼2. Fibres are flexible and elastic, with brittle failure and irregular fracture. No cleavage was observed. The mineral is readily soluble in cold H2O. The calculated density is 2.953 g cm−3. Optically, belakovskiite is biaxial (+) with α = 1.500(1), β = 1.511(1) and γ = 1.523(1) (measured in white light). The measured 2V is 87.1(6)° and the calculated 2V is 88°. The mineral is non-pleochroic. The partially determined optical orientation is X ≈ a. Electron-microprobe analysis provided Na2O 21.67, UO3 30.48, SO3 40.86, H2O 6.45 (structure), total 99.46 wt.% yielding the empirical formula Na6.83(U1.04O2)(SO4)4(S0.99O3OH)(H2O)3 based on 25 O a.p.f.u. Belakovskiite is triclinic, P
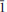 , with a = 5.4581(3), b = 11.3288(6), c = 18.4163(13) Å, α = 104.786(7)°, β = 90.092(6)°, γ = 96.767(7)°, V = 1092.76(11) Å3 and Z = 2. The eight strongest X-ray powder diffraction lines are [dobs Å(I)(hkl)]: 8.96(35)(002), 8.46(29)(011), 5.19(100)(
, with a = 5.4581(3), b = 11.3288(6), c = 18.4163(13) Å, α = 104.786(7)°, β = 90.092(6)°, γ = 96.767(7)°, V = 1092.76(11) Å3 and Z = 2. The eight strongest X-ray powder diffraction lines are [dobs Å(I)(hkl)]: 8.96(35)(002), 8.46(29)(011), 5.19(100)(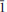 01,101,
01,101,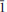 10), 4.66(58)(013,
10), 4.66(58)(013,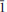 02,
02,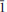
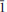 0,110), 3.568(37)(120,023,005,0
0,110), 3.568(37)(120,023,005,0 3), 3.057(59)(0
3), 3.057(59)(0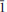 6,1
6,1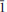 5,
5,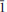 31), 2.930(27)(multiple) and 1.8320(29)(multiple). The structure, refined to R1 = 5.39% for 3163 Fo > 4σF reflections, contains [(UO2)(SO4)4(H2O)]6− polyhedral clusters connected via an extensive network of Na−O bonds and H bonds involving eight Na sites, three other H2O sites and an SO3OH (hydrosulfate) group. The 3-D framework, thus defined, is unique among known uranyl sulfate structures. The mineral is named for Dmitry Ilych Belakovskiy, a prominent Russian mineralogist and Curator of the Fersman Mineralogical Museum.
31), 2.930(27)(multiple) and 1.8320(29)(multiple). The structure, refined to R1 = 5.39% for 3163 Fo > 4σF reflections, contains [(UO2)(SO4)4(H2O)]6− polyhedral clusters connected via an extensive network of Na−O bonds and H bonds involving eight Na sites, three other H2O sites and an SO3OH (hydrosulfate) group. The 3-D framework, thus defined, is unique among known uranyl sulfate structures. The mineral is named for Dmitry Ilych Belakovskiy, a prominent Russian mineralogist and Curator of the Fersman Mineralogical Museum.
Bêhounekite, U(SO4)2(H2O)4, from Jáchymov (St Joachimsthal), Czech Republic: the first natural U4+ sulphate
- J. Plášil, K. Fejfarová, M. Novák, M. Dušek, R. Škoda, J. Hloušek, J. Čejka, J. Majzlan, J. Sejkora, V. Machovic, D. Talla
-
- Journal:
- Mineralogical Magazine / Volume 75 / Issue 6 / December 2011
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2739-2753
-
- Article
- Export citation
-
Bêhounekite, orthorhombic U(SO4)2(H2O)4, is the first natural sulphate of U4+. It was found in the Geschieber vein, Jáchymov (St Joachimsthal) ore district, Western Bohemia, Czech Republic, crystallized on the altered surface of arsenic and associated with kaatialaite, arsenolite, claudetite, unnamed phase UM1997-20-AsO:HU and gypsum. Bêhounekite most commonly forms short-prismatic to tabular green crystals, rarely up to 0.5 mm long. The crystals have a strong vitreous lustre and a grey to greenish grey streak. They are brittle with an uneven fracture and have very good cleavage along ﹛100﹜. The Mohs hardness is about 2. The mineral is not fluorescent either in short- or long-wavelength UV radiation. Bêhounekite is moderately pleochroic, α∼β is pale emerald green and γ is emerald green, and is optically biaxial (+) with α = 1.590(2), β = 1.618(4), γ = 1.659(2) (590 nm), 2V (calc.) = 81°, birefringence 0.069. The empirical formula of bêhounekite (based on 12 O atoms, from an average of five point analyses) is (U0.99Y0.03)Σ1.02(SO4)1.97(H2O)4. The simplified formula is U(SO4)2(H2O)4, which requires UO2 53.77. SO3 31.88, H2O 14.35, total 100.00 wt.%. Bêhounekite is orthorhombic, space group Pnma, a = 14.6464(3), b = 11.0786(3), c = 5.6910(14) Å, V = 923.43(4) Å3, Z = 4, Dcalc = 3.62 g cm–3. The seven strongest diffraction peaks in the X-ray powder diffraction pattern are [dobs in Å (I) (hid)]: 7.330 (100) (200), 6.112 (54) (210), 5.538 (21) (020), 4.787 (42) (111), 3.663 (17) (400), 3.478 (20) (410), 3.080 (41) (321). The crystal structure of bêhounekite has been solved by the charge-flipping method from single-crystal X-ray diffraction data and refined to R1 = 2.10 % with a GOF = 1.51, based on 912 unique observed diffractions. The crystal structure consists of layers built up from [8]-coordinate uranium atoms and sulphate tetrahedra. The eight ligands include four oxygen atoms from the sulphate groups and four oxygen atoms from the H2O molecules. Each uranium coordination polyhedron is connected via sulphate tetrahedra with other uranium polyhedra and through hydrogen bonds to the apices of sulphate tetrahedra. The dominant features of the Raman and infrared spectra of bêhounekite are related to stretching vibrations of SO4 tetrahedra (∼1200–950 cm–1), O-H stretching modes (∼3400–3000 cm–1) and H—O—H bending modes (∼1650 cm–1). The mineral is named in honour of František Bêhounek, a well known Czech nuclear physicist.
Leydetite, Fe(UO2)(SO4)2(H2O)11, a new uranyl sulfate mineral from Mas d’Alary, Lodève, France
- J. Plášil, A. V. Kasatkin, R. Škoda, M. Novák, A. Kallistová, M. Dušek, R. Skála, K. Fejfarová, J. Čejka, N. Meisser, H. Goethals, V. Machovič, L. Lapčák
-
- Journal:
- Mineralogical Magazine / Volume 77 / Issue 4 / June 2013
- Published online by Cambridge University Press:
- 05 July 2018, pp. 429-441
-
- Article
- Export citation
-
Leydetite, monoclinic Fe(UO2)(SO4)2(H2O)11(IMA 2012–065), is a new supergene uranyl sulfate from Mas d'Alary, Lodève, Hérault, France. It forms yellow to greenish, tabular, transparent to translucent crystals up to 2 mm in size. Crystals have a vitreous lustre. Leydetite has a perfect cleavage on (001). The streak is yellowish white. Mohs hardness is ∼2. The mineral does not fluoresce under long- or short-wavelength UV radiation. Leydetite is colourless in transmitted light, non-pleochroic, biaxial, with α = 1.513(2), γ = 1.522(2) (further optical properties could not be measured). The measured chemical composition of leydetite, FeO 9.28, MgO 0.37, Al2O30.26, CuO 0.14, UO340.19, SO321.91, SiO20.18, H2O 27.67, total 100 wt.%, leads to the empirical formula (based on 21 O a.p.f.u.), (Fe0.93Mg0.07Al0.04Cu0.01)Σ1.05(U1.01O2)(S1.96Si0.02)Σ1.98O8(H2O)11. Leydetite is monoclinic, space group C2/c, with a = 11.3203(3), b = 7.7293(2), c = 21.8145(8) Å, β = 102.402(3)°, V = 1864.18(10) Å3, Z = 4, and Dcalc = 2.55 g cm–3. The six strongest reflections in the X-ray powder diffraction pattern are [dobs in Å (I) (hkl)]: 10.625 (100) (002), 6.277 (1) (
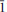 11), 5.321 (66) (004), 3.549 (5) (006), 2.663 (4) (008), 2.131 (2) (0 0 10). The crystal structure has been refined from single-crystal X-ray diffraction data to R1 = 0.0224 for 5211 observed reflections with [I > 3σ(I)]. Leydetite possesses a sheet structure based upon the protasite anion topology. The sheet consists of UO7 bipyramids, which share four of their equatorial vertices with SO4 tetrahedra. Each SO4 tetrahedron, in turn, shares two of its vertices with UO7 bipyramids. The remaining unshared equatorial vertex of the bipyramid is occupied by H2O, which extends hydrogen bonds within the sheet to one of a free vertex of the SO4 tetrahedron. Sheets are stacked perpendicular to the c direction. In the interlayer, Fe2+ ions and H2O groups link to the sheets on either side via a network of hydrogen bonds. Leydetite is isostructural with the synthetic compound Mg(UO2)(SO4)2(H2O)11. The name of the new mineral honours Jean Claude Leydet (born 1961), an amateur mineralogist from Brest (France), who discovered the new mineral.
11), 5.321 (66) (004), 3.549 (5) (006), 2.663 (4) (008), 2.131 (2) (0 0 10). The crystal structure has been refined from single-crystal X-ray diffraction data to R1 = 0.0224 for 5211 observed reflections with [I > 3σ(I)]. Leydetite possesses a sheet structure based upon the protasite anion topology. The sheet consists of UO7 bipyramids, which share four of their equatorial vertices with SO4 tetrahedra. Each SO4 tetrahedron, in turn, shares two of its vertices with UO7 bipyramids. The remaining unshared equatorial vertex of the bipyramid is occupied by H2O, which extends hydrogen bonds within the sheet to one of a free vertex of the SO4 tetrahedron. Sheets are stacked perpendicular to the c direction. In the interlayer, Fe2+ ions and H2O groups link to the sheets on either side via a network of hydrogen bonds. Leydetite is isostructural with the synthetic compound Mg(UO2)(SO4)2(H2O)11. The name of the new mineral honours Jean Claude Leydet (born 1961), an amateur mineralogist from Brest (France), who discovered the new mineral.
Meisserite, Na5(UO2)(SO4)3(SO3OH)(H2O), a new uranyl sulfate mineral from the Blue Lizard mine, San Juan County, Utah, USA
- J. Plášil, A. R. Kampf, A. V. Kasatkin, J. Marty, R. Škoda, S. Silva, J. Čejka
-
- Journal:
- Mineralogical Magazine / Volume 77 / Issue 7 / October 2013
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2975-2988
-
- Article
- Export citation
-
Meisserite (IMA2013-039), Na5(UO2)(SO4)3(SO3OH)(H2O), is a new uranyl sulfate mineral from the Blue Lizard mine, San Juan County, Utah (USA). It is named in honour of the prominent Swiss mineralogist Nicolas Meisser. The new mineral was found in a sandstone matrix and is associated with chalcanthite, copiapite, ferrinatrite, gypsum, johannite and another new Na-bearing uranyl sulfate, belakovskiite (IMA2013-075). Meisserite is a secondary mineral formed by the post-mining weathering of uraninite. The mineral is triclinic, P
 , a = 5.32317(10), b = 11.5105(2), c = 13.5562(10) Å, α = 102.864(7)°, β = 97.414(7)°, γ = 91.461(6)°, V = 801.74(6) Å3, and Z = 2. Crystals are prisms elongated on [100], up to 0.3 mm long, exhibiting the forms {010} and {001}. Meisserite is pale green to yellowish green, translucent to transparent and has a very pale yellow streak. It is brittle, with fair cleavage on {100} and {001}, and uneven fracture. The Mohs hardness is estimated at 2. Meisserite is somewhat hygroscopic and easily soluble in water. The calculated density based on the empirical formula is 3.208 g/cm3. Meisserite exhibits bright yellow green fluorescence under both long- and shortwave UV radiation. The mineral is optically biaxial (–), with α = 1.514(1), β = 1.546(1), γ = 1.557(1) (measured in white light). The measured 2V is 60(2)° and the calculated 2V is 60°. Dispersion is r > v, perceptible, and the optical orientation is X ≈ a, Z ≈ c*. The mineral is pleochroic, with X (colourless) < Y (pale yellow) ≈ Z (pale greenish yellow). The empirical formula of meisserite (based on 19 O a.p.f.u.) is Na5.05(U0.94O2)(SO4)3[SO2.69(OH)1.31](H2O). The Raman spectrum is dominated by the symmetric stretching vibrations of UO22+, SO42– and also weaker O–H stretching vibrations. The eight strongest powder X-ray diffraction lines are [dobs in Å (hkl)Irel]: 13.15 (001) 81, 6.33 (0
, a = 5.32317(10), b = 11.5105(2), c = 13.5562(10) Å, α = 102.864(7)°, β = 97.414(7)°, γ = 91.461(6)°, V = 801.74(6) Å3, and Z = 2. Crystals are prisms elongated on [100], up to 0.3 mm long, exhibiting the forms {010} and {001}. Meisserite is pale green to yellowish green, translucent to transparent and has a very pale yellow streak. It is brittle, with fair cleavage on {100} and {001}, and uneven fracture. The Mohs hardness is estimated at 2. Meisserite is somewhat hygroscopic and easily soluble in water. The calculated density based on the empirical formula is 3.208 g/cm3. Meisserite exhibits bright yellow green fluorescence under both long- and shortwave UV radiation. The mineral is optically biaxial (–), with α = 1.514(1), β = 1.546(1), γ = 1.557(1) (measured in white light). The measured 2V is 60(2)° and the calculated 2V is 60°. Dispersion is r > v, perceptible, and the optical orientation is X ≈ a, Z ≈ c*. The mineral is pleochroic, with X (colourless) < Y (pale yellow) ≈ Z (pale greenish yellow). The empirical formula of meisserite (based on 19 O a.p.f.u.) is Na5.05(U0.94O2)(SO4)3[SO2.69(OH)1.31](H2O). The Raman spectrum is dominated by the symmetric stretching vibrations of UO22+, SO42– and also weaker O–H stretching vibrations. The eight strongest powder X-ray diffraction lines are [dobs in Å (hkl)Irel]: 13.15 (001) 81, 6.33 (0 2) 62, 5.64 (0
2) 62, 5.64 (0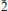 1,020) 52, 5.24 (100,012,
1,020) 52, 5.24 (100,012, 01) 100, 4.67 (101) 68, 3.849 (
01) 100, 4.67 (101) 68, 3.849 (
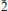 1,102,022) 48, 3.614 (0
1,102,022) 48, 3.614 (0 2,
2,
 3) 41, and 3.293 (
3) 41, and 3.293 ( 13,004) 43. The crystal structure of meisserite (R1 = 0.018 for 3306 reflections with Iobs > 3σI) is topologically unique among known structures of uranyl minerals and inorganic compounds. It contains uranyl pentagonal bipyramids linked by SO4 groups to form chains. Na+ cations bond to O atoms in the chains and to an SO3OH group and an H2>O group between the chains, thereby forming a heteropolyhedral framework.
13,004) 43. The crystal structure of meisserite (R1 = 0.018 for 3306 reflections with Iobs > 3σI) is topologically unique among known structures of uranyl minerals and inorganic compounds. It contains uranyl pentagonal bipyramids linked by SO4 groups to form chains. Na+ cations bond to O atoms in the chains and to an SO3OH group and an H2>O group between the chains, thereby forming a heteropolyhedral framework.
The crystal chemistry of the uranyl carbonate mineral grimselite, (K, Na)3Na[(UO2)(CO3)3](H2O), from Jáchymov, Czech Republic
- J. Plášil, K. Fejfarová, R. Skála, R. Škoda, N. Meisser, J. Hlousek, I. Císařová, M. Dušek, F. Veselovský, J. Čejka, J. Sejkora, P. Ondruš
-
- Journal:
- Mineralogical Magazine / Volume 76 / Issue 3 / June 2012
- Published online by Cambridge University Press:
- 05 July 2018, pp. 443-453
-
- Article
- Export citation
-
Two crystals of the uranyl carbonate mineral grimselite, ideally K3Na[(UO2)(CO3)3](H2O), from Jáchymov in the Czech Republic were studied by single-crystal X-ray diffraction and electron-probe microanalysis. One crystal has considerably more Na than the ideal chemical composition due to substitution of Na into KO8 polyhedra; the composition of the other crystal is nearer to ideal, and similar to synthetic grimselite. The presence of Na atoms in KO8 polyhedra, which are located in channels in the crystal structure, reduces their volume, and as a result the unit-cell volume also decreases. Structure refinement shows that the formula for the sample with the anomalously high Na content is (K2.43Na0.57)Σ3.00Na[(UO2)(CO3)3](H2O). The unit-cell parameters, refined in space group P
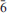 2c, are a = 9.2507(1), c = 8.1788(1) Å, V = 606.14(3) Å3 and Z = 2. The crystal structure was refined to R1 = 0.0082 and wR1 = 0.0185 with a GOF = 1.33, based on 626 observed diffraction peaks [Iobs>3σ(I)].
2c, are a = 9.2507(1), c = 8.1788(1) Å, V = 606.14(3) Å3 and Z = 2. The crystal structure was refined to R1 = 0.0082 and wR1 = 0.0185 with a GOF = 1.33, based on 626 observed diffraction peaks [Iobs>3σ(I)].
Calciodelrioite, Ca(VO3)2(H2O)4, the Ca analogue of delrioite, Sr(VO3)2(H2O)4
- A. R. Kampf, J. Marty, B. P. Nash, J. Plášil, A. V. Kasatkin, R. Škoda
-
- Journal:
- Mineralogical Magazine / Volume 76 / Issue 7 / December 2012
- Published online by Cambridge University Press:
- 05 July 2018, pp. 2803-2817
-
- Article
- Export citation
-
Calciodelrioite, ideally Ca(VO3)2(H2O)4, is a new mineral (IMA 2012-031) from the uraniumvanadium deposits of the eastern Colorado Plateau in the USA. The type locality is the West Sunday mine, Slick Rock district, San Miguel County, Colorado. The new mineral occurs on fracture surfaces in corvusite- and montroseite-impregnated sandstone and forms as a result of the oxidative alteration of these phases. At the West Sunday mine, calciodelrioite is associated with celestine, gypsum, huemulite, metarossite, pascoite and rossite. The mineral occurs as transparent colourless needles, bundles of tan to brown needles and star bursts of nearly black broad blades composed of tightly intergrown needles. Crystals are elongate and striated parallel to [100], exhibiting the prismatic forms {001} and {011} and having terminations possibly composed of the forms {100} and {61
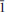 }. The mineral is transparent and has a white streak, subadamantine lustre, Mohs hardness of about 2½, brittle tenacity, irregular to splintery fracture, one perfect cleavage on {001} and possibly one or more additional cleavages parallel to [100]. Calciodelrioite is soluble in water. The calculated density is 2.451 g cm– 3. It is optically biaxial (+) with α = 1.733(3), β = 1.775(3), γ = 1.825(3) (white light), 2Vmeas = 87.3(9)° and 2Vcalc = 87°. The optical orientation is X = b; Z ≈ a. No pleochroism was observed. Electronmicroprobe analyses of two calciodelrioite samples and type delrioite provided the empirical formulae (Ca0.88Sr0.07Na0.04K0.01)Σ1.00(V1.00O3)2(H2.01O)4, (Ca0.76Sr0.21Na0.01)Σ0.98(V1.00O3)2(H2.01O)4 and (Sr0.67Ca0.32)Σ0.99(V1.00O3)2(H2.00O)4, respectively. Calciodelrioite is monoclinic, I2/a, with unit-cell parameters a = 14.6389(10), b = 6.9591(4), c = 17.052(2) Å, β = 102.568(9)°, V = 1695.5(3) Å3 and Z = 8. The seven strongest lines in the X-ray powder diffraction pattern [listed as dobs Å (I)(hkl)] are as follows: 6.450(100)(011); 4.350(16)(013); 3.489(18)(020); 3.215(17)(022); 3.027(50)(multiple); 2.560(28)(
}. The mineral is transparent and has a white streak, subadamantine lustre, Mohs hardness of about 2½, brittle tenacity, irregular to splintery fracture, one perfect cleavage on {001} and possibly one or more additional cleavages parallel to [100]. Calciodelrioite is soluble in water. The calculated density is 2.451 g cm– 3. It is optically biaxial (+) with α = 1.733(3), β = 1.775(3), γ = 1.825(3) (white light), 2Vmeas = 87.3(9)° and 2Vcalc = 87°. The optical orientation is X = b; Z ≈ a. No pleochroism was observed. Electronmicroprobe analyses of two calciodelrioite samples and type delrioite provided the empirical formulae (Ca0.88Sr0.07Na0.04K0.01)Σ1.00(V1.00O3)2(H2.01O)4, (Ca0.76Sr0.21Na0.01)Σ0.98(V1.00O3)2(H2.01O)4 and (Sr0.67Ca0.32)Σ0.99(V1.00O3)2(H2.00O)4, respectively. Calciodelrioite is monoclinic, I2/a, with unit-cell parameters a = 14.6389(10), b = 6.9591(4), c = 17.052(2) Å, β = 102.568(9)°, V = 1695.5(3) Å3 and Z = 8. The seven strongest lines in the X-ray powder diffraction pattern [listed as dobs Å (I)(hkl)] are as follows: 6.450(100)(011); 4.350(16)(013); 3.489(18)(020); 3.215(17)(022); 3.027(50)(multiple); 2.560(28)(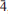 15,413); 1.786(18)(028). In the structure of calciodelrioite (refined to R1 = 3.14% for 1216 Fo > 4σF), V5+O5 polyhedra link by sharing edges to form a zigzag divanadate [VO3] chain along a, similar to that in the structure of rossite. The chains are linked via bonds to Ca atoms, which also bond to H2O groups, yielding CaO3(H2O)6 polyhedra. The Ca polyhedra form a chain along b. Each of the two symmetrically independent VO5 polyhedra has two short vanadyl bonds and three long equatorial bonds. Calciodelrioite and delrioite are isostructural and are the endmembers of the series Ca(VO3)2(H2O)4–Sr(VO3)2(H2O)4. Calciodelrioite is dimorphous with rossite, which has a similar structure; however, the smaller 8-coordinate Ca site in rossite does not accommodate Sr.
15,413); 1.786(18)(028). In the structure of calciodelrioite (refined to R1 = 3.14% for 1216 Fo > 4σF), V5+O5 polyhedra link by sharing edges to form a zigzag divanadate [VO3] chain along a, similar to that in the structure of rossite. The chains are linked via bonds to Ca atoms, which also bond to H2O groups, yielding CaO3(H2O)6 polyhedra. The Ca polyhedra form a chain along b. Each of the two symmetrically independent VO5 polyhedra has two short vanadyl bonds and three long equatorial bonds. Calciodelrioite and delrioite are isostructural and are the endmembers of the series Ca(VO3)2(H2O)4–Sr(VO3)2(H2O)4. Calciodelrioite is dimorphous with rossite, which has a similar structure; however, the smaller 8-coordinate Ca site in rossite does not accommodate Sr.
Vysokýite, U4+[AsO2(OH)2]4·4H2O, a new mineral from Jáchymov, Czech Republic
- J. Plášil, J. Hloušek, R. Škoda, M. Novák, J. Sejkora, J. Čejka, F. Veselovský, J. Majzlan
-
- Journal:
- Mineralogical Magazine / Volume 77 / Issue 8 / December 2013
- Published online by Cambridge University Press:
- 05 July 2018, pp. 3055-3066
-
- Article
- Export citation
-
Vysokýite, U4+[(AsO2(OH)2]4(H2O)4 (IMA 2012–067), was found growing on an altered surface of massive native As in the Geschieber vein, Jáchymov ore district, Western Bohemia, Czech Republic. The new mineral was found in association with běhounekite, štěpite, kaatialaite, arsenolite, claudetite and gypsum. It forms extremely fibrous light-green crystals up to 8 mm long. Crystals have an alabaster lustre and a greenish-white to greyish streak. Vysokýite is brittle with uneven fracture and perfect cleavage along (100) and (001); the Mohs hardness is ∼2. A density of 3.393 g/cm3 was calculated using the empirical formula and unit-cell parameters obtained from a single-crystal diffraction experiment. Vysokýite is non-fluorescent under short or long wavelength UV radiation. It is colourless under the microscope, measured refractive indices are α' = 1.617(3), γ' = 1.654(3); the estimated optical orientation is α' ∼X, γ' ∼Z. The average of five spot wavelength dispersive spectroscopy (WDS) analyses is 29.44 UO2, 1.03 SiO2, 48.95 As2O5, 0.12 SO3, 15.88 H2O (calc.), total 95.42 wt.%. The empirical formula of vysokýite (based on 20 O a.p.f.u.) is U1.00[AsO2(OH)2]3.90(SiO4)0.16 (SO4)0.01·4H2O. The As–O–H and O–H vibrations dominate in the Raman spectrum. Vysokýite is triclinic, space group P
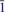 , with a = 10.749(2), b = 5.044(3), c = 19.1778(7) Å, α = 89.872(15)°, β = 121.534(15)°, γ = 76.508(15)°, and V = 852.1(6) Å3, Z = 2 and Dcalc = 3.34 g·cm–3. The strongest diffraction peaks in the X-ray powder diffraction pattern are [dobs in Å (Irel.)(hkl)]: 8.872(100)(100), 8.067(50)(002), 6.399(7)(10
, with a = 10.749(2), b = 5.044(3), c = 19.1778(7) Å, α = 89.872(15)°, β = 121.534(15)°, γ = 76.508(15)°, and V = 852.1(6) Å3, Z = 2 and Dcalc = 3.34 g·cm–3. The strongest diffraction peaks in the X-ray powder diffraction pattern are [dobs in Å (Irel.)(hkl)]: 8.872(100)(100), 8.067(50)(002), 6.399(7)(10 ), 4.773(6)(10
), 4.773(6)(10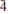 ), 3.411(10)(30
), 3.411(10)(30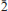 ), 3.197(18)(31
), 3.197(18)(31 ). The crystal structure of vysokýite was solved from single-crystal X-ray diffraction data by the charge-flipping method and refined to R1 = 0.0595 based on 2718 unique observed reflection, and to wR2 = 0.1160 for all 4173 unique reflections. The structure of vysokýite consists of UO8 square antiprisms sharing all of their vertices with 8 As-tetrahedra to form infinite chains parallel to [010]. These chains are linked by hydrogen bonds involving terminal (OH) groups of the double-protonated As-tetrahedra and molecules of H2O located between the chains. The new mineral is named in honour of Arnošt Vysoký (1823–1872), the former chief of the Jáchymov mines and smelters, chemist and metallurgist.
). The crystal structure of vysokýite was solved from single-crystal X-ray diffraction data by the charge-flipping method and refined to R1 = 0.0595 based on 2718 unique observed reflection, and to wR2 = 0.1160 for all 4173 unique reflections. The structure of vysokýite consists of UO8 square antiprisms sharing all of their vertices with 8 As-tetrahedra to form infinite chains parallel to [010]. These chains are linked by hydrogen bonds involving terminal (OH) groups of the double-protonated As-tetrahedra and molecules of H2O located between the chains. The new mineral is named in honour of Arnošt Vysoký (1823–1872), the former chief of the Jáchymov mines and smelters, chemist and metallurgist.




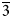
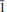 , with
, with 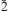 02)(74), 2.9783 (
02)(74), 2.9783 (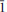 21)(74) and 2.7231 (112)(31). The crystal structure of nestolaite was determined by means of the Rietveld refinement from the powder data to
21)(74) and 2.7231 (112)(31). The crystal structure of nestolaite was determined by means of the Rietveld refinement from the powder data to 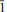 , with a = 8.7434(11), b = 8.309(3), c = 8.8693(10) Å , a = 77.86(2), b = 104.635(11), g = 82.935(18)°, V = 598.8(3) A˚
, with a = 8.7434(11), b = 8.309(3), c = 8.8693(10) Å , a = 77.86(2), b = 104.635(11), g = 82.935(18)°, V = 598.8(3) A˚ 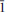 , with
, with 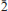 0); 3.097(49)(1
0); 3.097(49)(1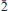 01); 3.791(26)(
01); 3.791(26)(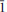 21), 2.968(22)(
21), 2.968(22)(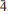 01); for cobaltoblödite: 4.551(80)(210, 011); 4.269(50)(
01); for cobaltoblödite: 4.551(80)(210, 011); 4.269(50)(
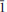 , with
, with  3), 3.057(59)(0
3), 3.057(59)(0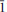 11), 5.321 (66) (004), 3.549 (5) (006), 2.663 (4) (008), 2.131 (2) (0 0 10). The crystal structure has been refined from single-crystal X-ray diffraction data to
11), 5.321 (66) (004), 3.549 (5) (006), 2.663 (4) (008), 2.131 (2) (0 0 10). The crystal structure has been refined from single-crystal X-ray diffraction data to  ,
, 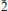 1,020) 52, 5.24 (100,012,
1,020) 52, 5.24 (100,012, 2,
2,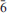 2
2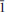 }. The mineral is transparent and has a white streak, subadamantine lustre, Mohs hardness of about 2½, brittle tenacity, irregular to splintery fracture, one perfect cleavage on {001} and possibly one or more additional cleavages parallel to [100]. Calciodelrioite is soluble in water. The calculated density is 2.451 g cm
}. The mineral is transparent and has a white streak, subadamantine lustre, Mohs hardness of about 2½, brittle tenacity, irregular to splintery fracture, one perfect cleavage on {001} and possibly one or more additional cleavages parallel to [100]. Calciodelrioite is soluble in water. The calculated density is 2.451 g cm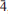 15,413); 1.786(18)(028). In the structure of calciodelrioite (refined to R
15,413); 1.786(18)(028). In the structure of calciodelrioite (refined to R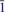 , with
, with  ), 4.773(6)(10
), 4.773(6)(10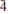 ), 3.411(10)(30
), 3.411(10)(30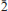 ), 3.197(18)(31
), 3.197(18)(31